Developing novel treatments for cancer
- Most cancer therapies are not curative in advanced cancer patients
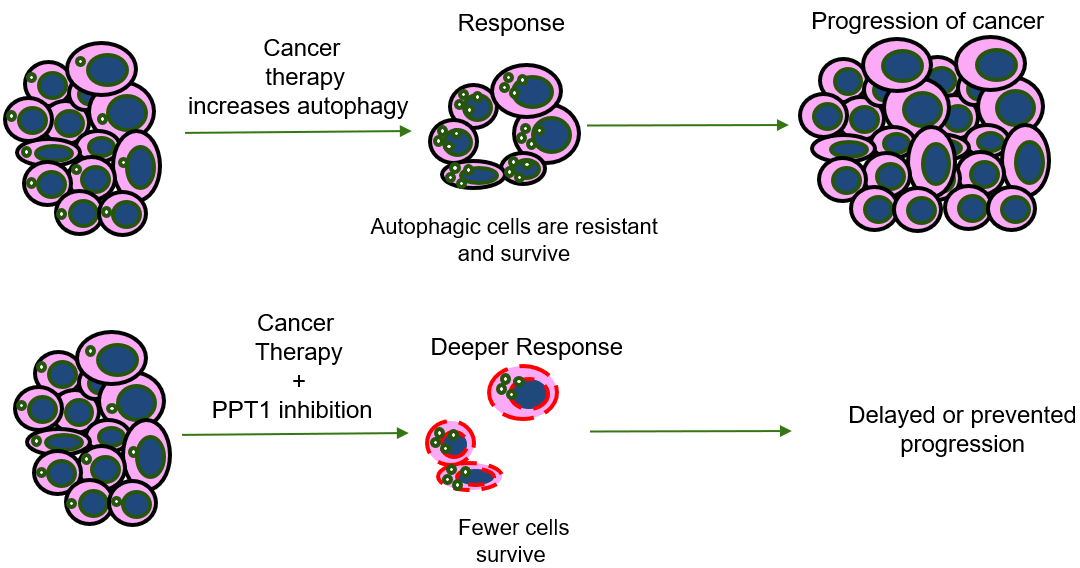
Some cancer therapies never produce a response in the patient (primary resistance) - Even if cancer therapies produce a response, the response is often temporary
- Some cancer cells survive that go on to produce progression (regrowth of tumor)
- Therapy resistance is a major contributor to cancer deaths
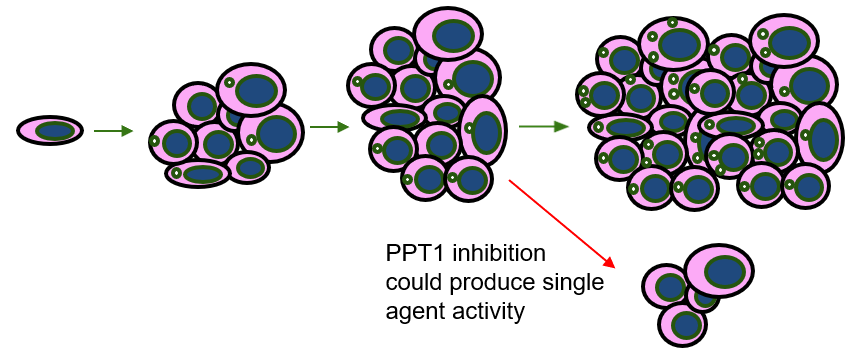
- Many cancers rely on autophagy to resist treatment, but efforts to block autophagy and overcome therapy resistance have not been successful thus far
- Pinpoint’s innovative technology focuses on a new target in the autophagy pathway, the PPT1 enzyme, that offers an exciting new approach to overcoming cancer therapy resistance
- Pinpoint’s inhibitors provide the chemical innovation that increases the potency and specificity of targeting PPT1 and the autophagy pathway
- We anticipate Pinpoint PPT1 inhibitors will be effective in multiple cancers that have been shown to rely on the autophagy pathway
Our Vision: A new class of treatment for therapy-resistant cancers
Compelling reasons Pinpoint’s PPT1 inhibitors could be practice-changing for multiple cancers
- Either genetic or chemical PPT1 inhibition shows significant tumor growth impairment in vivo in multiple models.
- In most cancers, PPT1 expression is higher in cancer versus normal tissue, and in metastases versus primary tumors
- Pinpoint has developed potent and specific PPT1 inhibitors
- Pinpoint inhibitors produce
- Single agent antitumor activity
- Combine well with other agents
- Enhance immunotherapy, targeted therapy, and chemotherapy in vivo
- Hydroxychloroquine is a first generation PPT1 inhibitor, used safely in thousands of patients worldwide, that augments the efficacy of cancer therapy in colon cancer, pancreas cancer and melanoma clinical trials
- Existing data suggest that Pinpoint’s innovative technology may enhance efficacy as compared to hydroxychloroquine, a first-generation PPT1 inhibitor, used safely in thousands of patients worldwide, that augments the efficacy of cancer therapy in multiple clinical trials.
About Us
Pinpoint Therapeutics is developing novel cancer treatment options aimed to specifically inhibit autophagy, a key cell survival and treatment resistance pathway in cancer. Pinpoint inhibitors target a recently discovered enzyme in the autophagy pathway called PPT1, which is highly expressed across most cancers. These inhibitors could be applied to a broad range of cancers alone or in combination to overcome therapy resistance. Further development of these compounds towards clinical drug candidates will be undertaken by Pinpoint Therapeutics to determine the clinical utility of this novel treatment strategy.
Pinpoint Team

Christian Peters, MD, PhD
CEO
Joining Pinpoint in 2019, Christian brings with him 28+ years of successful scientific, clinical, development and commercialization experience in both academia and the pharmaceutical industry, including CSL Behring, J&J spinoff Therakos and start-up Realm Therapeutics. Instrumental in obtaining FDA approval for seven products.

Jeffrey Winkler, PhD
Co-Founder
Merriam Professor of Chemistry, Founding Member, Penn Center for Cancer Pharmacology; Member, Abramson Cancer Center; University of Pennsylvania.
Organic and medicinal chemistry expertise.

Ravi Amaravadi, MD
Co-Founder
Associate Professor of Medicine, Co-Leader Cancer Therapeutics Program – Abramson Cancer Center, University of Pennsylvania.
Autophagy expertise, Clinical Trialist.
Key Publications
“Autophagy and cancer”: >13,000 papers
Examples: Lum Cell 2005; Dagenhardt Cancer Cell 2006; Yang Genes & Development 2011, Guo Genes & Development 2013, Strohecker Cancer Discovery 2013, Xie Cancer Discovery 2014, Yang Cancer Discovery 2014; Karsli Uzunbas Cancer Discovery 2014; Sousa Nature 2016; Yang Cancer Discovery 2018; Poillet-Perez Nature 2018
“Chloroquine and cancer”: >2000 papers
Examples: Ma J. Clin Inv 2014; Muclahy Levy Cancer Discovery 2014; Perera Nature 2016; Morgan PNAS 2018; Kinsey Nature Medicine 2019; Bryant Nature Medicine 2019; Luo PNAS 2019.
Amaravadi/Winkler Lab Publications
Mcafee PNAS 2012; Ma JCI 2014; Piao Autophagy 2017; Rebecca Cancer Discovery 2017; Rebecca Cancer Discovery 2019
Other groups that have used dimeric chloroquines
Pelligrini Autophagy 2014- Sweden; Eng PNAS 2015- Pfizer/Novartis; Ndoye Cancer Res 2017- Wistar; Devorkin Mol Can Res 2017-Canada; Frazier Cancer Res 2017- Presage Biosciences; Gade Radiology 2017 – Penn; Whelan Oncogene 2017- Penn; Vijayaraghavan Nat. Comm. 2017- MD Anderson; Baquero Leukemia 2018-Ireland; CechakovaInt J Mol Sci. 2019- Czech republic ; Santiago-O’Farrill JM Cancer 2019 – MD Anderon
Review Article: Amaravadi Cancer Discovery 2019